1. Introduction
2. Experiment Details
2.1 Materials
Table┬Ā1
| Position | 1G |
| Polarity | DCEP |
| Current | 125-110 A |
| Voltage | 20-25 V |
| Speed | 1.6 mm/sec |
| Electrode | E308BRN/E309 Žå4mm |
Table┬Ā2
2.2 Test Procedures
Table┬Ā3
Fig.┬Ā1

3. Results
3.1 Microstructural Characterization
3.1.1 Base metal microstructure
3.1.2 Weld metal Microstructure
Fig.┬Ā3
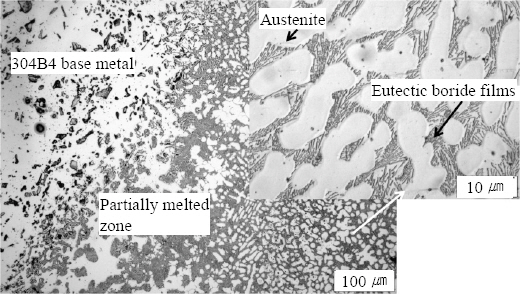
Fig.┬Ā4

Fig.┬Ā5

3.2 Solidification simulation
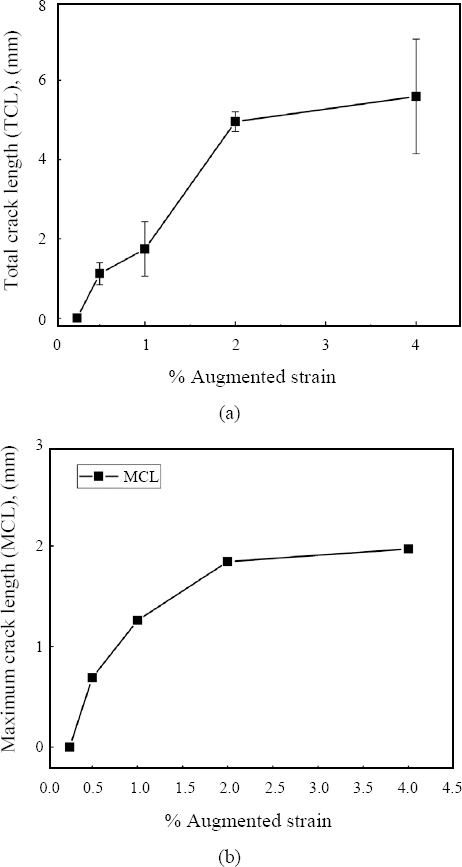
3.3 Hot cracking susceptibility
3.4 Mechanical Properties
3.4.1 Tensile properties
3.4.2 Impact properties
Fig.┬Ā11

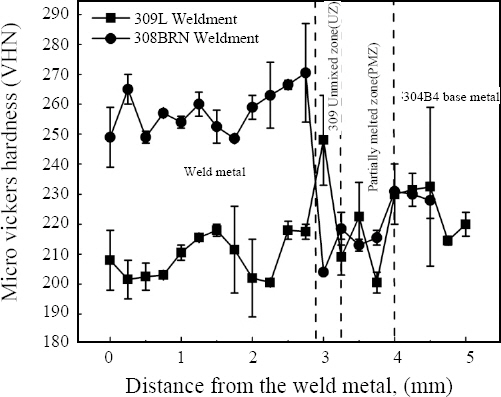
3.4.3 Microhardness Survey across the weldments
4. Discussion
4.1 Hot cracking susceptibility of E308BRN weld metal
4.2 Comparison of mechanical properties of E308BRN and E309 weld joint
5. Conclusion
1) Boron containing E308BRN weld joint is found to be moderately susceptible for solidification cracking during multipass welding due to ineffective backfilling by Fe-rich eutectic borides in long and wide cracks. However, E308BRN electrodes can be used for applications involving low to moderate restraint forces during welding to produce crack free joints.
2) Tensile properties of both E309 and E308 weld joints are poor due to the presence of eutectic borides in the interdendritic regions of E309 weld metal diluted by base metal in and intentional addition of B in case of E308BRN.
3) E309 weld joint consist of more or less uniform strength across the weldment though microstructures across the weldment are not uniform.
4) E308BRN weld joint shows large mismatch in strength between the weld metal and base metal and hence failed in a brittle manner without any ductility. Although, the electrode has similar composition as that of the base metal.
5) Poor impact toughness observed in E308BRN weld metal is attributed to as cast structure of the weld metal consisting of continuous network of eutectic borides along the interdendritic regions. Whereas, good impact toughness shown by E309 weld metal is due to presence of only austenite - ferrite duplex microstructure and no traces of eutectic boride.













 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print