1. Introduction
Many types of automobile sheet steels have been studied to acquire an increasing demand; high strength and elongation. Development of quenching and partitioning (Q&P) steels is also underway to satisfy the demand. In modern times, Q&P process is applied to ferritic and martensitic stainless steels1, 2). The Q&P steel is normally composed of carbon-depleted martensite and carbon-enriched retained austenite. The hard microstructure (martensite) is produced by the initial quenching to a temperature between martensite-start (Ms) and martensite-finish (Mf) temperatures following the full austenization or intercritical annealing. And, it is subsequently heat-treated through two different processes: (i) holding at the initial quenching temperature (one step Q&P), (ii) partitioning at a higher temperature than the initial quenching temperature (two step Q&P)3). Consequently, the Q&P steel exhibits transformation-induced plasticity (TRIP) phenomenon due to the existence of carbon-enriched and stabilized austenite. Bainite and new martensite can be obtained from the unstabilized austenite during the partitioning and the final quenching step, respectively4, 5). Furthermore, the Q&P steels produced the carbide precipitates delaying/prohibiting the stabilization of the retained austenite6).
There are the other phase transformations observed for the Q&P steels such as isothermal martensite transformation7, 8) and bainitic ferrite transformation9). These phase transformations influence the amount and stabilization of the retained austenite. The stability of the retained austenite was studied in a 0.09C steel by measuring the variation of new Ms temperature10). The isothermal martensite transformation occurs normally in high carbon steels11). For a low carbon (0.15C wt.%) Q&P steel, Kim et al. found the isothermal martensite transformation/bainite transformation as a function of partitioning temperature and time12). However, the isothermal martensite and bainite transformation were not exactly defined in their study. Specifically, the changes of microstructures are not investigated with the stability of retained austenite for Q&P steels.
The objective of the study is to identify the isothermal martensite/bainite transformations and its effect on the retained austenite during the partitioning (one step Q&P and two step Q&P) processes by measuring a dilatation and microstructural behavior. Furthermore, this study will be important to understand and estimate the microstructural evolution of the weldment and heat affected zone13, 14).
2. Experimental procedure
A cold-rolled steel sheet was used for phase transformation analysis. The steel had a composition of Fe-0.15C-1.48Si-1.5Mn (wt.%). Heat treatment conditions were selected by considering Ac1, Ac3, and Ms temperatures which were estimated from the available equations15, 16). The Ac1 and Ac3 temperatures were mostly the same as compared with Thermo-Calc software. An intercritical annealing temperature to have a phase fraction (╬▒ : ╬│ = 25 : 75) was set to 885┬░C using Thermo-Calc software.
An initial quenching temperature was determined to produce the most retained austenite by Koistinen ŌĆō Marburger relationship17):
where fm is the volume fraction of a martensite produced from the initial quenching, QT is the initial quenching temperature, and %╬│ is the volume fraction of an austenite at the intercritical annealing temperature. Dilatometry specimens were fabricated to the size (1├Ś3├Ś10mm) and the intercritical annealing was executed at 885┬░C for 270 seconds, followed by quenching to the room temperature (cooling rate ~ 40┬░C/s). Dilatation conditions for phase transformation analysis are shown in Table 1. Ht denotes the holding time following the initial quenching, PT the partitioning temperature, and Pt the partitioning time. Heating and cooling rates for Q&P dilatation test were the same as the continuous cooling dilatation. The microstructures of the samples were investigated using light optical microscopy (LOM) and scanning electron microscopy (SEM). The samples used in the LOM and SEM analyses were etched with 2% nital and sodium metabisulfite (Na2S2O5 + H2O 100 ml).
3. Result and discussion
Fig. 1 shows the dilatation behavior of continuous heating and cooling from the intercritical annealing temperature (885┬░C). An epitaxial ferrite (red circle in Fig. 1a) and a martensite (blue circle in Fig. 1a) were formed from the austenite during quenching at 630~780┬░C and 101~300┬░C, respectively. It is normally assumed that a para-equilibrium state is maintained during quenching from the intercritical annealing18). The para-equilibrium during quenching permits transfer of interstitial alloy elements and restricts transfer of substitutional alloy elements. During the intercritical annealing step, the slight variation of substitutional concentration may occur in the austenite. Therefore, the epitixial ferrite was produced in some austenites during quenching. Due to the formation of the epitaxial ferrite, the volume fraction of intercritical austenites should decrease and the carbon content in the intercritical austenite should increase. The measured Ms (300┬░C) was lower than the calculated Ms (403┬░C), which was conformed as the carbon content in the intercritical austenite increased.
Fig.┬Ā1
Dilitation of continuous cooling from intercritical annealing (885┬░C); (a) epitaxial ferrite and martensite transformations, (b) an enlarged section of martensite transformation
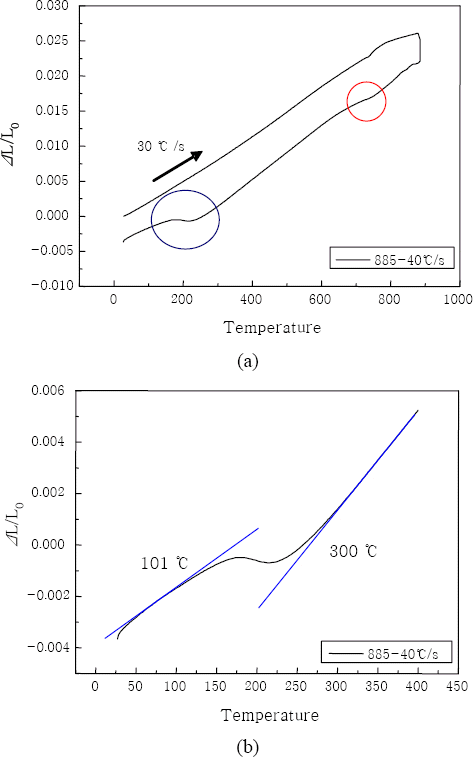
Fig. 2a shows the representative section of martensite transformation for the one-step Q&P dilatation (holding for 30s). The Ms temperature (298┬░C) was mostly same as that for the continuous cooling dilatation (300┬░C). The length increment was observed during the holding period at 240┬░C post to the initial quenching. During the last quenching following the holding process, a new Ms temperature was observed and new martensite was produced from the unstable austenite. Speer et al. also found the new Ms in the Q&P steel19). Fig. 2b shows the length increment and the new Ms decrease as a function of holding time. A dilatometer used in the study needed a duration (less than 1 second) for thermal stabilization in the beginning of holding process, which was not included in the analysis. The length increment during the holding (240┬░C) was probably due to the isothermal martensite transformation. The isothermal martensite transformation could occur because the holding temperature was between Ms and Mf. In the beginning of the holding process, the isothermal martensite was significantly produced because the holding temperature was lower than Ms (298┬░C). As the holding time increased, the new Ms decreased since the carbon partitioning from the carbon-enriched martensite to the retained austenite increased the stability of the retained austenite. Therefore, the isothermal bainitic transformation could also occur on the holding of 240┬░C because the holding temperature was larger than the new Ms in this study. The isothermal martensite/bainite transformation was retarded as the holding time was increased and as the discrepancy between the new Ms and the holding temperature was increased. A carbon enrichment of the retained austenite during the holding process played a role in decreasing the driving force of the isothermal martensite transformation.
Fig.┬Ā2
Dilatation for one step Q&P process (initial quenching to 240┬░C, followed by holding 30s at 240┬░C and the final quenching); (a) Ms obtained from the initial quenching and new Ms from the final quenching, (b) a length increment and new Ms decrease with respect to the holding time

Fig. 3a shows the representative section of martensite transformation for the two-step Q&P dilatation (holding 5s at 240┬░C, followed by partitioning at 350┬░C for 1000s). The Ms temperature (296┬░C) was mostly same as that for the continuous cooling dilatation (300┬░C). The length increment was observed during the holding period at 240┬░C and the partitioning period at 350┬░C. During the final quenching following the partitioning process, a new Ms was observed and new martensite was produced from the unstable austenite. However, as the partitioning time increased, the new Ms decreased and disappeared completely when the partitioning at 350┬░C proceeded for 1000s following the holding 5s at 240┬░C (Fig. 3a). Increasing the partitioning time, the carbon enrichment in the retained austenite should increase, therefore enhancing the stability of the retained austenite and reducing the Ms.
Fig.┬Ā3
Dilatation for two step Q&P process (initial quenching to 240┬░C, followed by holding 5s at 240┬░C, by partitioning at 350 and 400┬░C, and by the final quenching); (a) Ms obtained from the initial quenching and new Ms disappeared for 1000s partitioning, (b) a length increment and new Ms decrement with respect to the partitioning time

Fig. 3b shows the length increment and the new Ms decrement as a function of partitioning time. An isothermal length increment generated for the partitioning step was probably due to bainite transformation because the partitioning temperature was higher than the Ms and new Ms. The isothermal bainite transformation indicated a different behavior as compared with the isothermal martensite transformation: The length increment was minimal in the beginning of the partitioning time. However, as the partitioning proceeded to ~30s, the dilatation showed a significant increment (Fig. 3b). The partitioning time to ~30s might be considered as the bainite incubation period20). On the other hand, the isothermal martensite transformation was mostly produced for the holding time less than 10s (Fig. 2b).
In case of the partitioning executed at 400┬░C post to the holding at 240┬░C, the new Ms dropped more quickly than the case of 350┬░C partitioning (Fig. 3b). The partitioning at 400┬░C should acquire the larger thermal driving force for carbon partitioning than the 350┬░C partitioning. Therefore, the 400┬░C partitioning indicated the shorter incubation period for the bainite transformation than the 350┬░C partitioning. The isothermal bainite transformation was retarded as the partitioning time was increased to ~400s. A quick drop of Ms and short period of bainite incubation for the 400┬░C partitioning steel were the direct evidence of significant effects of carbon partitioning on the stability of retained austenite21).
Detailed SEM microstructures for one step and two step Q&P steels are depicted in Fig. 4. Blocky martensite shape is certainly appearing as initial quenching, holding process and the final quenching (Fig. 4a). As holding time increased, fine martensite of lath type in blocky martensite increased. The authors suppose that fine martensite of lath type is formed from untransformed austenite by isothermal transformation during holding process. Consequently, the final microstructure is composed to athermal martensite by initial quenching, isothermal martensite/bainite by holding and new martensite (athermal martensite) by the final quenching.
Fig.┬Ā4
Microstructure of Q&P steels as a function of heat treatment conditions: (a,e) 30s holding at 240┬░C, (b,f) 10s partitioning at 350┬░C post to holding 5s at 240┬░C, (c,g) 1000s partitioning at 350┬░C post to holding 5s at 240┬░C, (d,h) 5s partitioning at 400┬░C post to holding 5s at 240┬░C
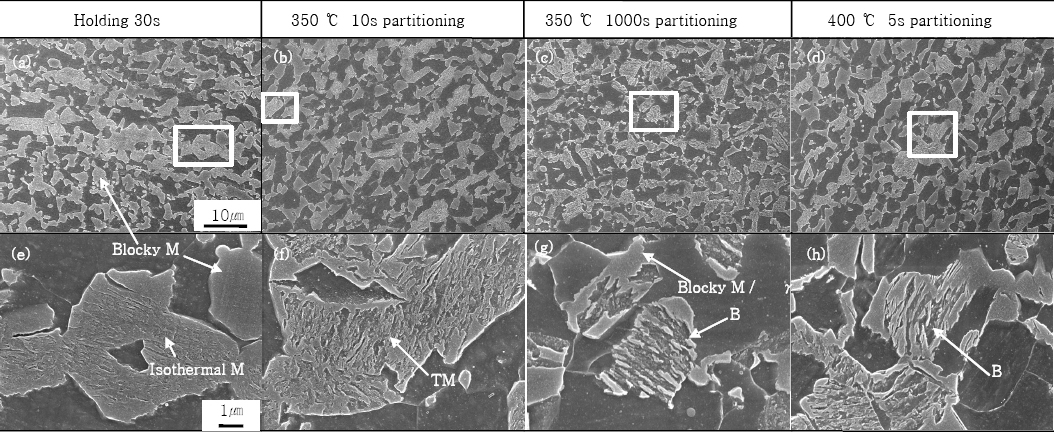
Blocky martensite of 350┬░C partitioned specimen decreased in contrast with the specimens partitioned at 240┬░C. This is due to that remained untransformed austenite was transformed to bainite during partitioning at 350┬░C. As partitioning time decreased, blocky martensite and tempered martensite were more frequently observed for 350┬░C partitioning process. In contrast, as partitioning time increased, bainite shape increased (Fig. 4c). As the stability of retained austenite increased by carbon partitioning, athermal martensite transformation decreased during the final quenching.
In case of 400┬░C partitioning, bainite transformation occurred even at 5s partitioned specimen (Fig. 4d). This is because bainite incubation period effect is disappeared and more rapidly approach to bainite transformation area as partitioning temperature increases. And blocky martensite quantity decreased as compared with 10s partitioning for 350┬░C. The new martensite transformation decreased as retained austenite stability was more rapidly progressed by carbon partitioning in case of partitioning at higher temperature. Consequently, the final microstructure is composed to athermal martensite by initial quenching, isothermal martensite/bainite by holding process, bainite by partitioning process and new martensite (athermal martensite) by the final quenching.
The study was focused on the phase transformation from the retained austenite to martensite/bainite during the partitioning (one step Q&P and two step Q&P) processes with no direct observation of mechanical properties. The amount and stability of retained austenite have an interactive, sometimes independent, relationship with the Q&P-process condition such as QT, Ht, PT, and Pt21, 22). Therefore, it is very difficult to estimate the tensile properties directly from the dilatation conditions (QT, Ht, PT, and Pt), as shown in Table 1. The effects of the Q&P conditions on the mechanical property will be discussed in future study associated with the phase transformation and microstructural behavior.
4. Conclusion
Isothermal martensite/bainite transformation occurred during partitioning period for 0.15C quenching and partitioning (Q&P) steels. Dilatation for the one step Q&P process was conducted by initial quenching to 240┬░C, followed by holding 30s at 240┬░C and the final quenching. Carbon partitioning from supersaturated martensite to untransformed austenite was conducted at the partitioning process and new martensite was formed by the final quenching. The isothermal martensite transformation occurred because the holding temperature was between Ms and Mf. The isothermally transformed martensite/bainite and the athermally transformed martensite were produced by a loss of retained austenite. Dilatation for the two step Q&P process was conducted by initial quenching to 240┬░C, followed by holding 5s at 240┬░C, by partitioning at 350 and 400┬░C, and by the final quenching. The isothermal bainitic transformation occurred because the partitioning temperature was larger than the Ms. The partitioning at 400┬░C indicated the short incubation period for the bainite transformation than the 350┬░C partitioning. The partitioning at 400┬░C had the larger thermal driving force for carbon partitioning than the 350┬░C partitioning. The isothermally transformed martensite/bainite, a quick drop of Ms, and short period of bainite incubation for the 400┬░C partitioning steel were the direct evidence of significant effects of carbon partitioning on the stability of retained austenite.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print